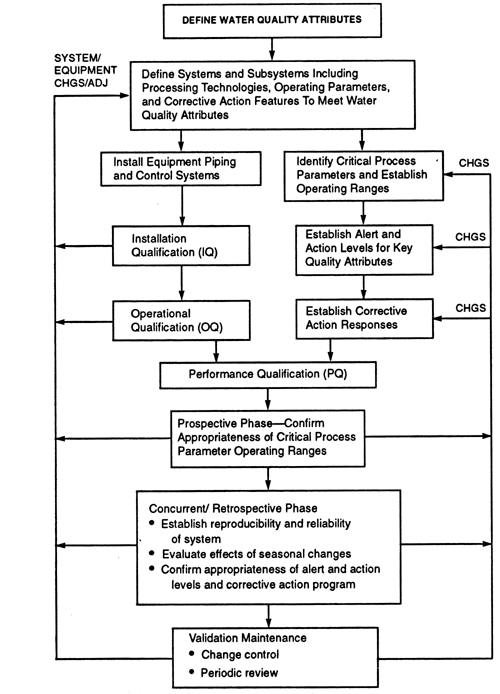
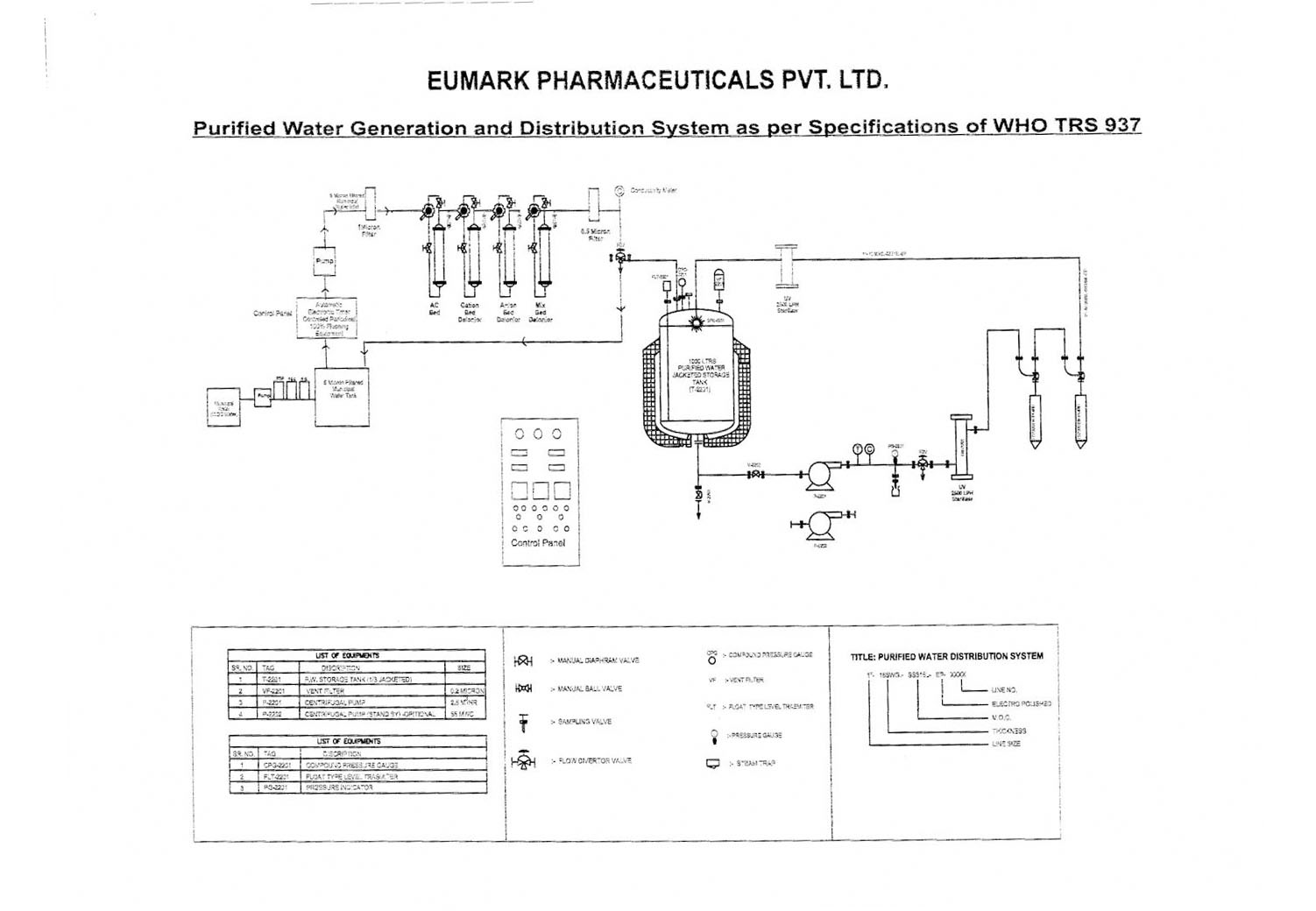
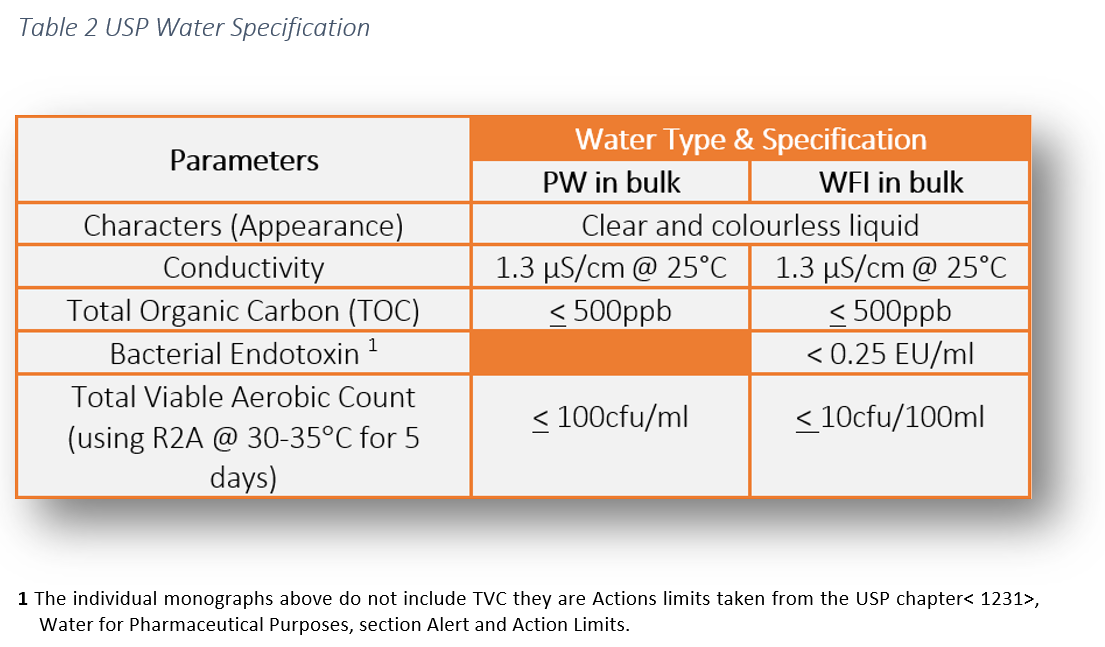
Microbial Monitoring of Pharmaceutical Grade Water Systems – Common Misconceptions | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology
OmniPur® Water, WFI Quality, Sterile Purified Water, Cell Culture Tested - CAS 7732-18-5 - Calbiochem | 4.86505
CHEMICAL QUALITY CONTROL ON WATER PRODUCED FROM WFI SYSTEM AT MEDICAL TECHNOLOGY DIVISION, NUCLEAR MALAYSIA. Muhammad Hanaffi Mo

Comparison of WFI production by membrane based method & distillation based method, according to the Revised EP Monograph for WFI Production - A3P - Pharmaceutical & Biotechnology Industry




![Purified Water/ Feed Water Specification [5]. | Download Scientific Diagram Purified Water/ Feed Water Specification [5]. | Download Scientific Diagram](https://www.researchgate.net/publication/350711835/figure/tbl1/AS:1021794060353536@1620626096413/Purified-Water-Feed-Water-Specification-5_Q320.jpg)