Consumption of CO 2 and production of HCO 3 − in silicates and calcium... | Download Scientific Diagram
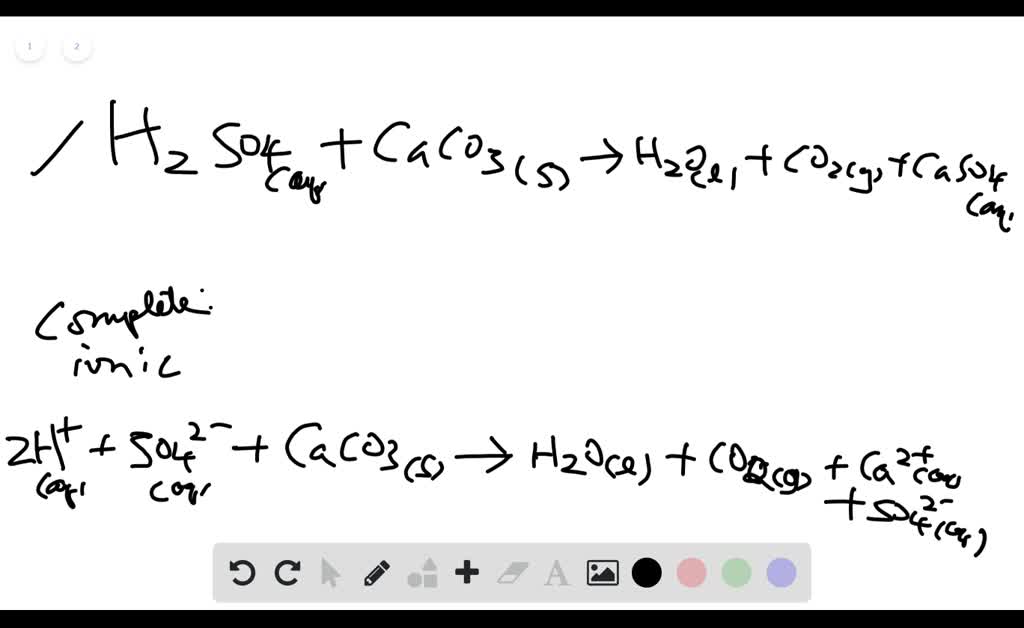
SOLVED: Write complete ionic and net ionic equations for the reaction between sulfuric acid (H2 SO4) and calcium carbonate (Ca CO3) . H2 SO4(aq)+ CaCO3(s) →H2 O(l)+CO2(g)+CaSO4(aq) | Numerade
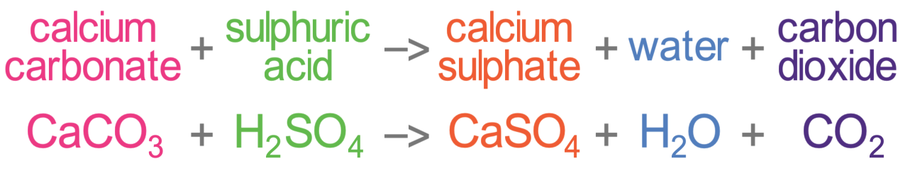
What is calcium carbonate + sulfuric acid as a word equation and a balanced symbol equation? - Quora
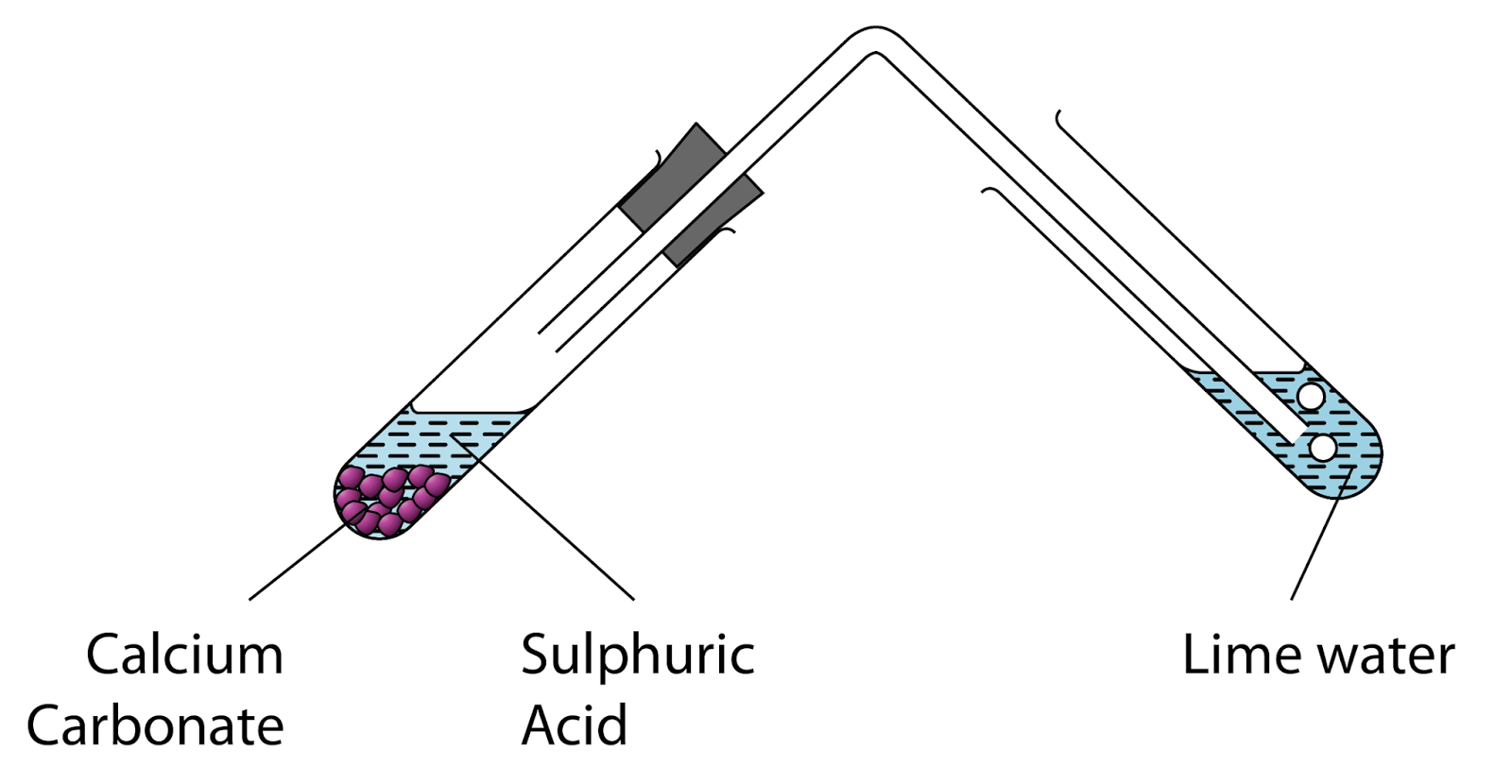
reactivity - How could mass increase when sulfuric acid is added to calcium carbonate? - Chemistry Stack Exchange

28.5 g of calcium carbonate (CaCOn reacts with an excess of sulfuric acid (H SOg) to farm calcium sulfate - brainly.com

SOLVED: One of the results of acid rain is the reaction of sulfuric acid, H2SO4, with calcium carbonate, CaCO3, to produce calcium sulfate, CaSO4, and carbonic acid, H2CO3. The chemical equation for