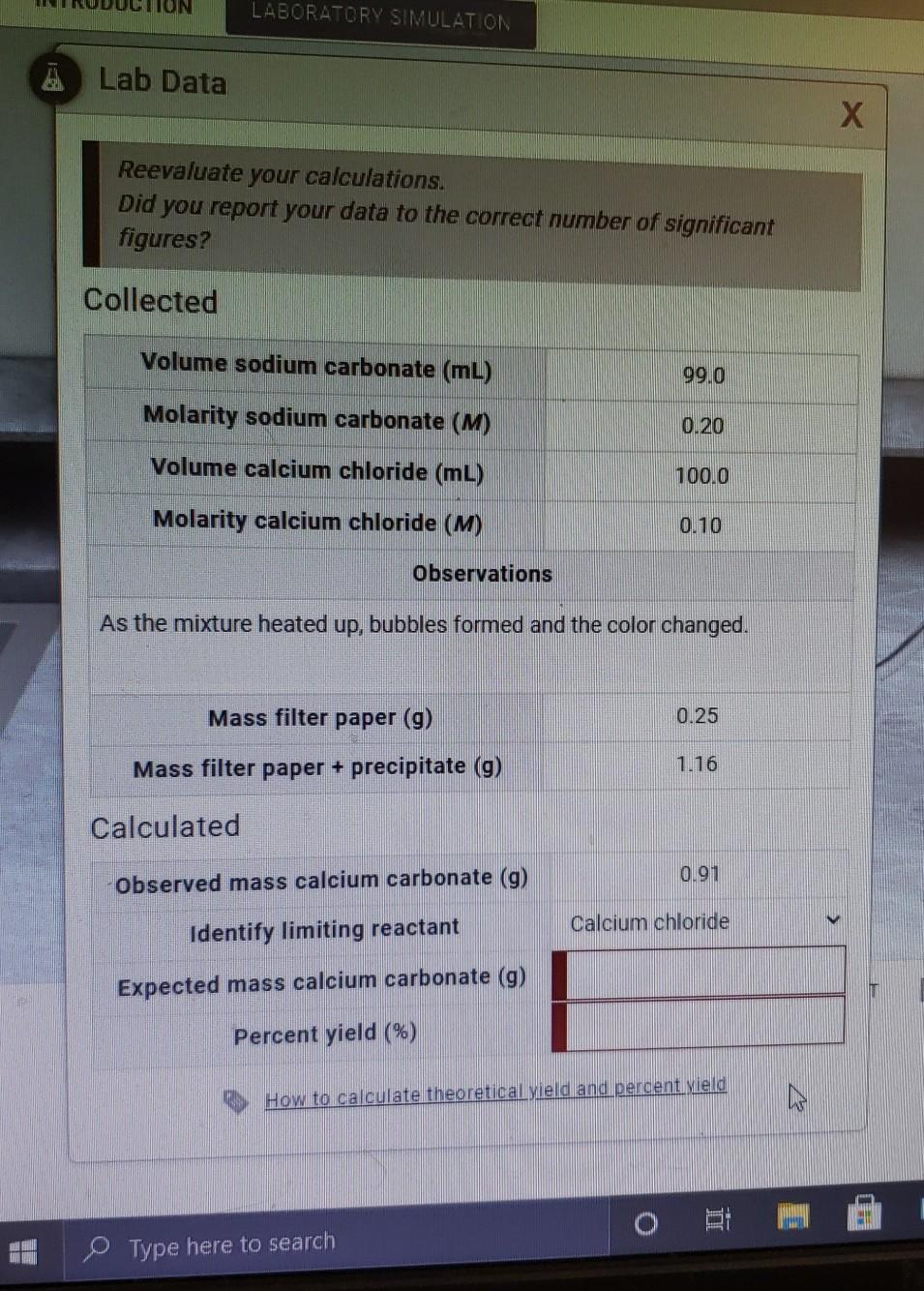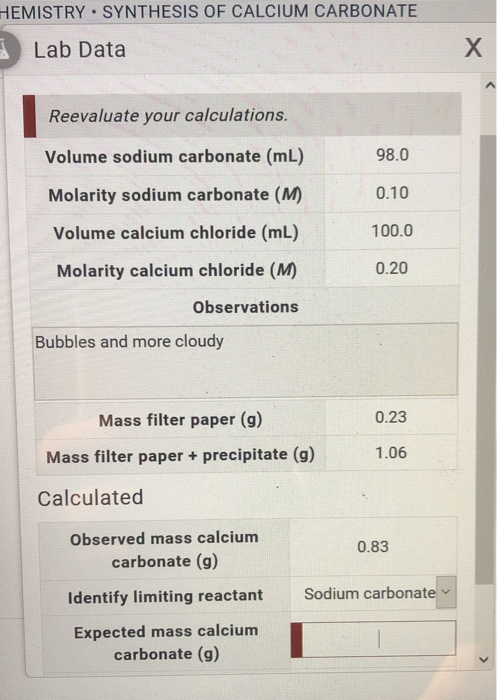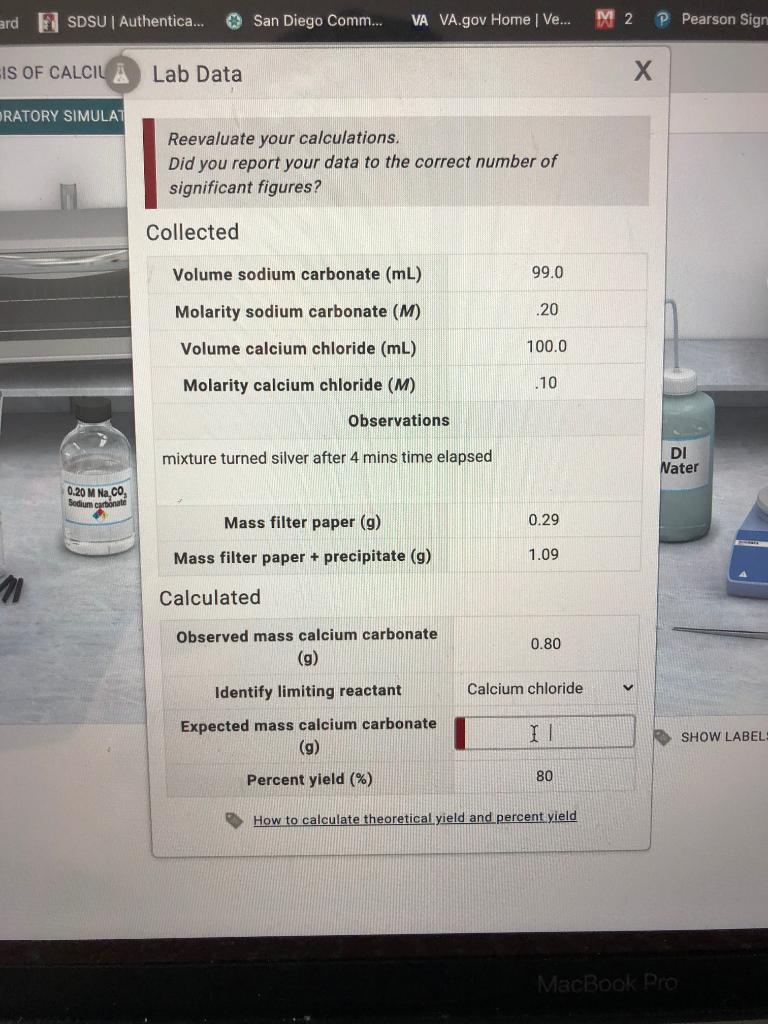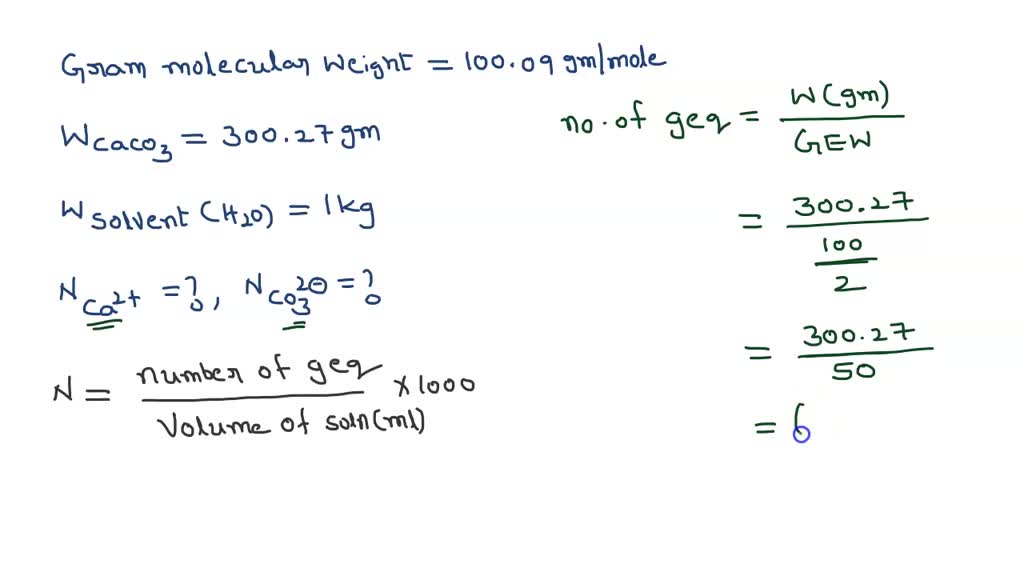
SOLVED: The molecular mass of CaCO3 is 100.09 g/mol. If 300.27 g of CaCO3 is dissolved in 1 kg of pure water, what is the normality (in eq/kg of Ca2+ and CO3
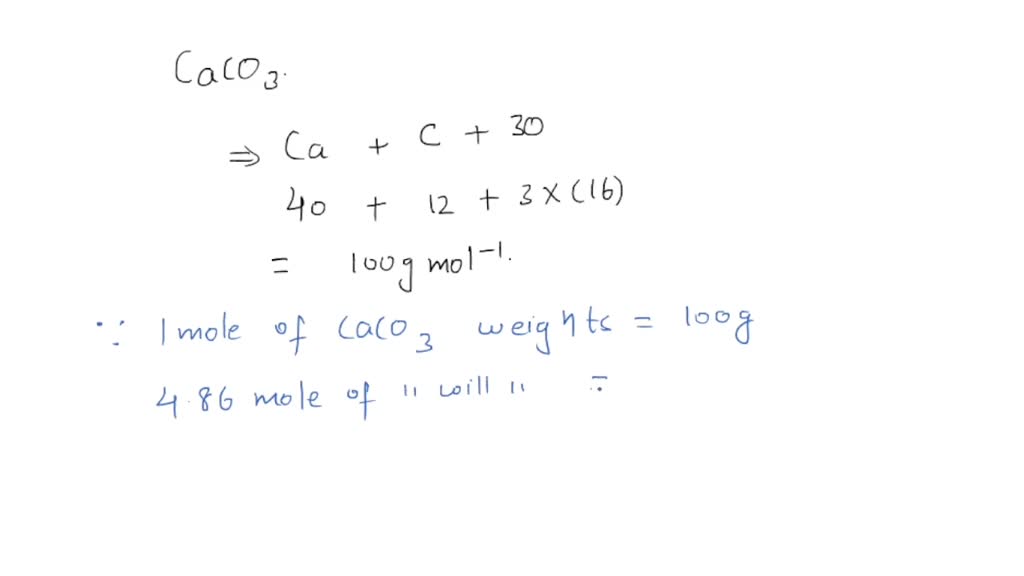
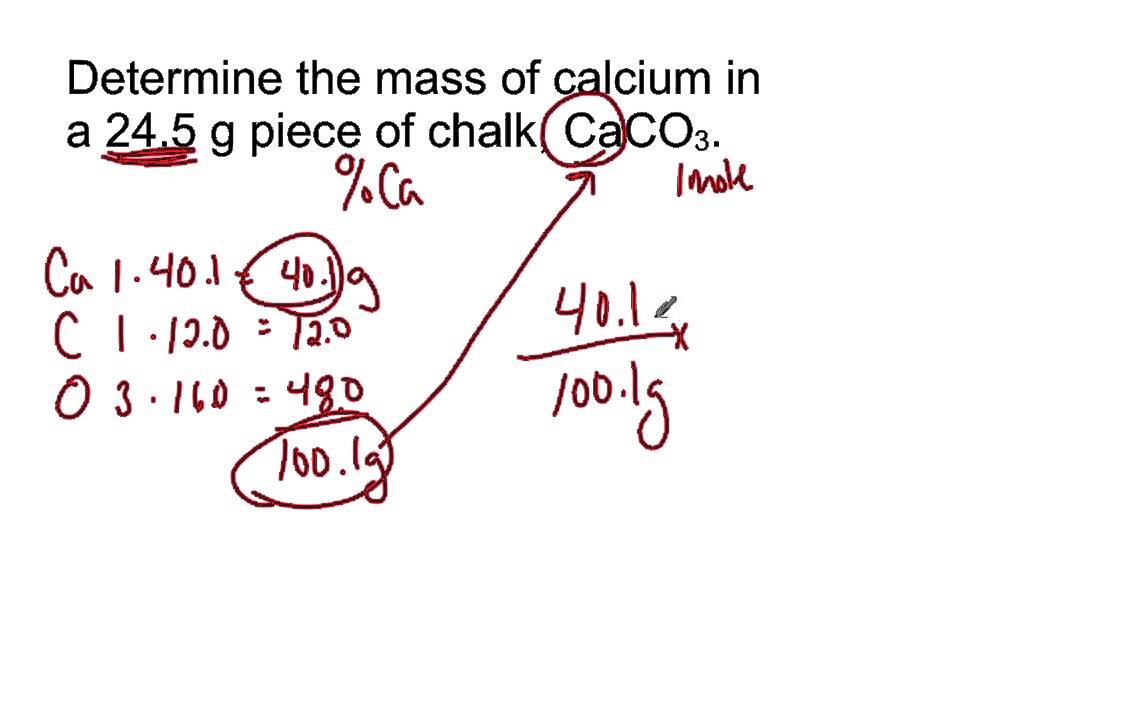
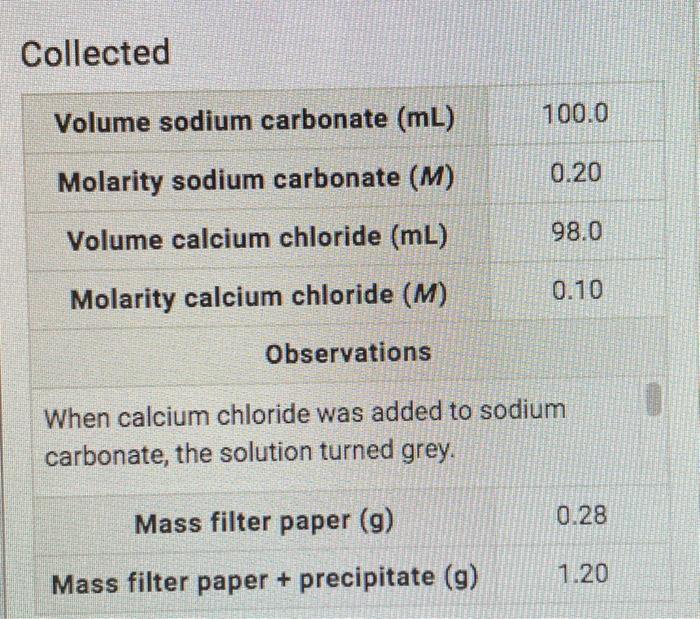
SOLVED: Calculating Molar Mass II Calcium carbonate (CaCO;). also called calcite, is the principal mineral found in lime- stone, marble. chalk; pearls, and the shells of marine animals such as clams Calculate
Law of conservation of mass - Chemistry calculations - Edexcel - GCSE Chemistry (Single Science) Revision - Edexcel - BBC Bitesize
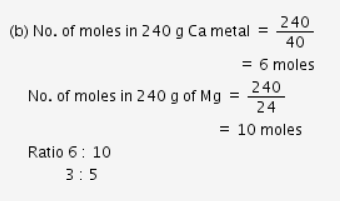
Calculate the molecular mass of CaCO3 (At mass Ca = 40 u, C = 12 u, O = 16 u) - CBSE Class 9 - Learn CBSE Forum
Law of conservation of mass - Chemistry calculations - Edexcel - GCSE Chemistry (Single Science) Revision - Edexcel - BBC Bitesize
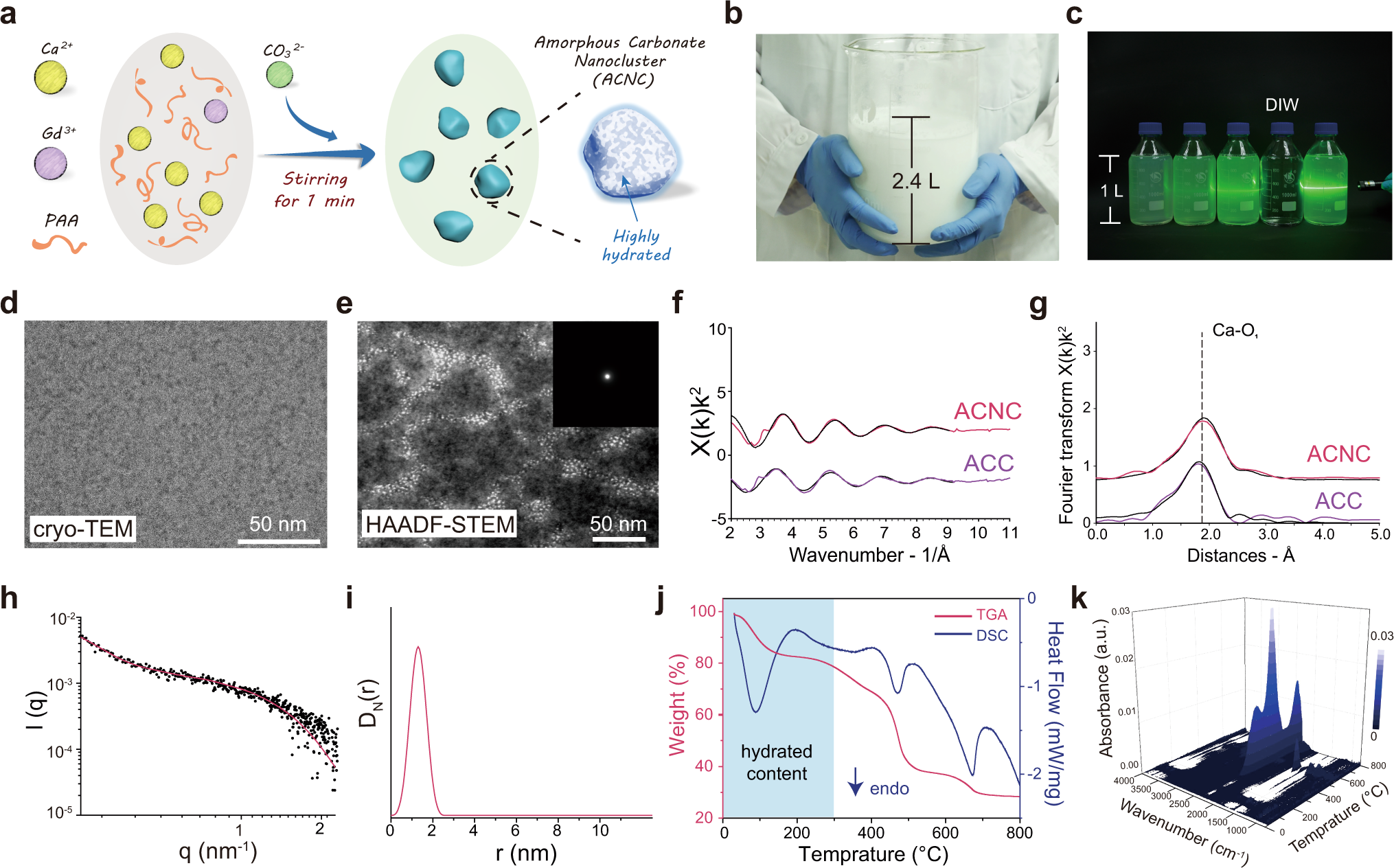
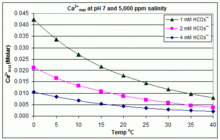
Highly hydrated paramagnetic amorphous calcium carbonate nanoclusters as an MRI contrast agent | Nature Communications