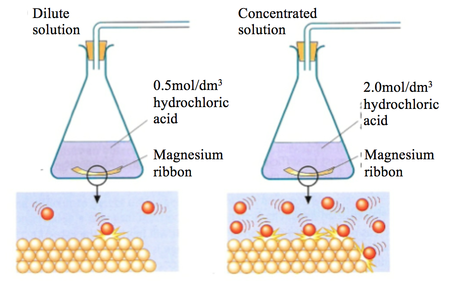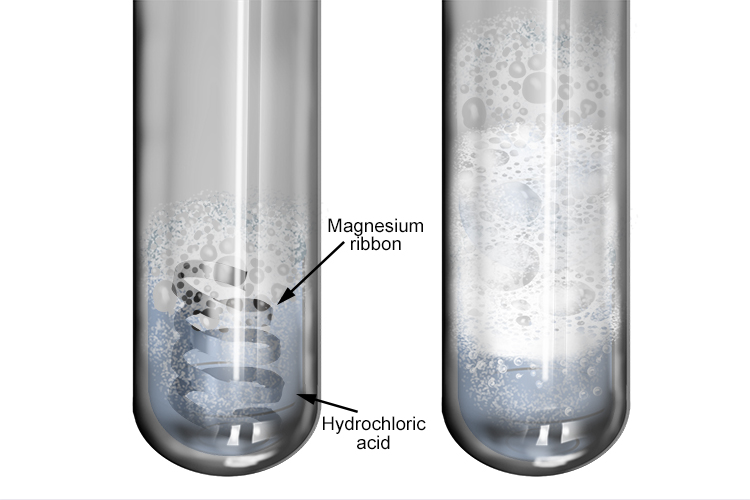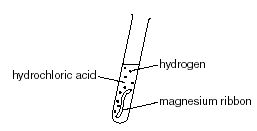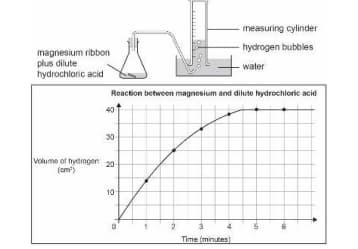
A piece of magnesium ribbon is added to a flask containing dilute hydrochloric acid Hydrogen gas is formed which is collected in the measuring cylinder The line on the graph indicates the
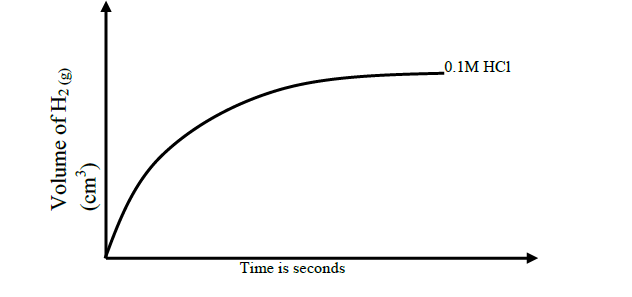
The sketch of the graph below shows a reaction between magnesium ribbon and dilute hydrochloric acid.

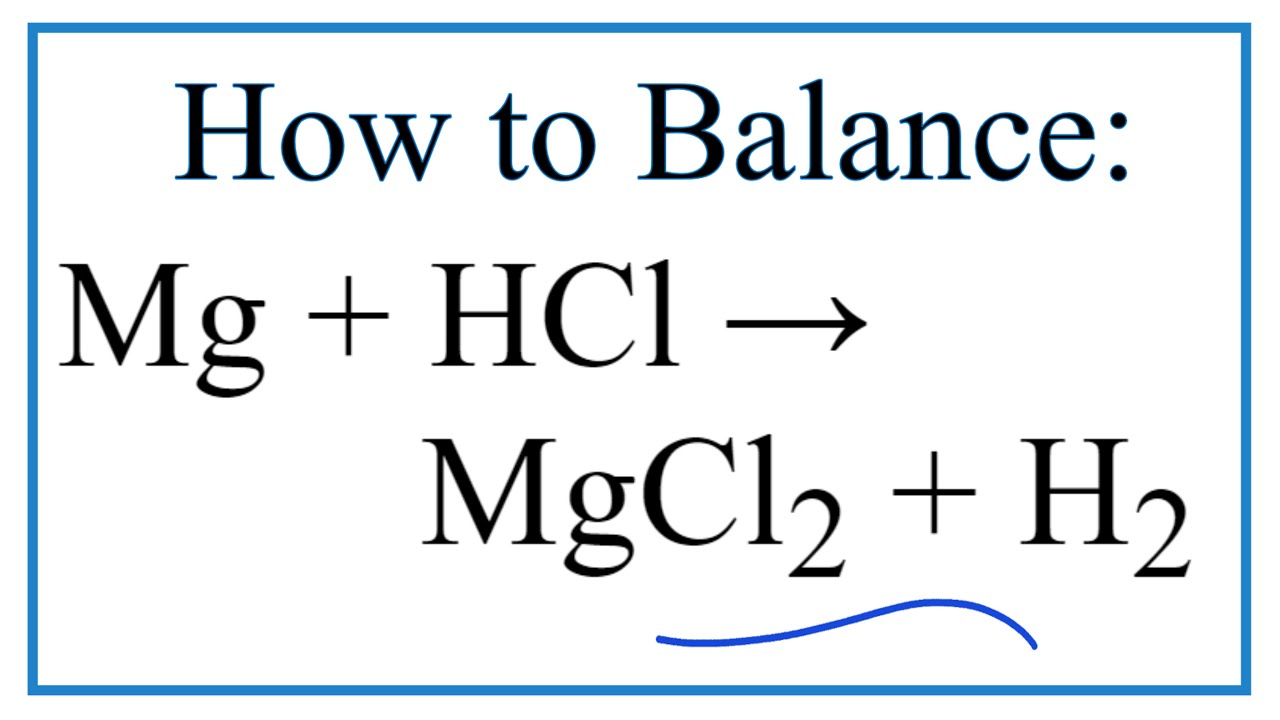
Bildagentur | mauritius images | Magnesium (Mg) ribbon in a beaker reacting with hydrochloric acid (HCl). This reaction creates magnesium chloride (MgCl2) and hydrogen gas (H2). Mg + 2HCl --> MgCl2 + H2

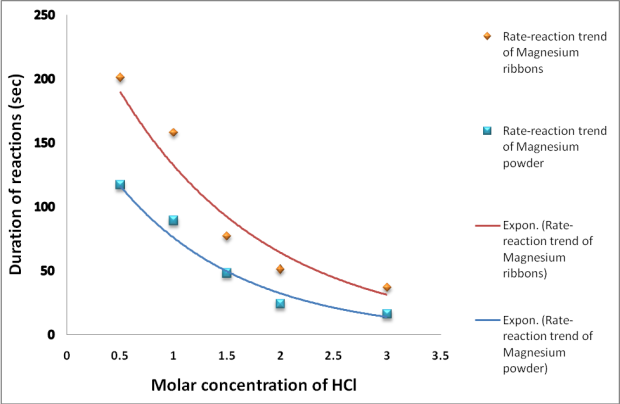
Describe an experiment to study the speed of reaction between dilute hydrochloric acid and magnesium, by measuring the volume of gas produced over time. - Study notes, tips, worksheets, exam papers