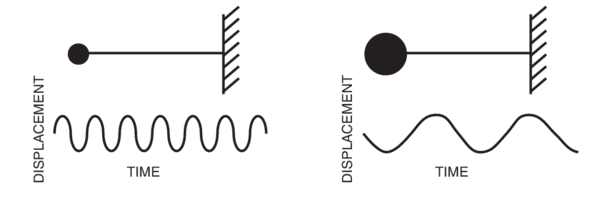
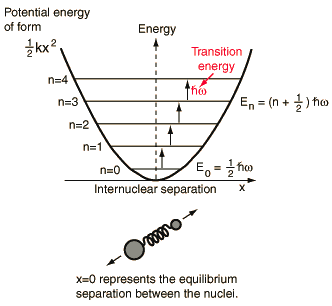
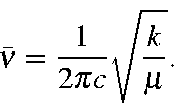For H2 molecule, the fundamental vibrational frequency can be taken as 4400 cm-1. The zeropoint energy of the molecule is ______ kJ mol-1 (rounded up to two decimal places).[h = 6.6andtimes;10-34 J

SOLVED: The fundamental vibrational frequency of the H2 molecule is 4200 cm-1. Calculate the energy level spacing and the ZPE, in J and in kJ/mol. h = 1.0 x 10^-34 J·s, c =

OneClass: Consider a diatomic molecule such as HCl. The fundamental vibrational frequency has been me...

Calculation of Vibrational Frequency - Introduction to Organic Chemistry - Lecture Notes | Study notes Organic Chemistry | Docsity

Equilibrium vibration frequency, Anharmonicity, Zero point Energy, Force Constant – IR Spectroscopy - YouTube