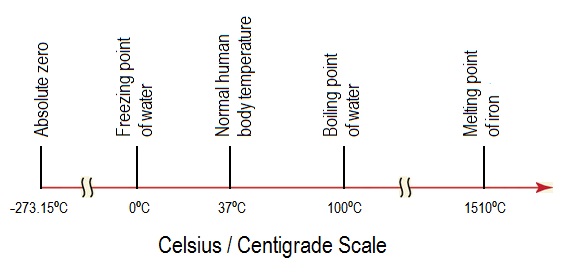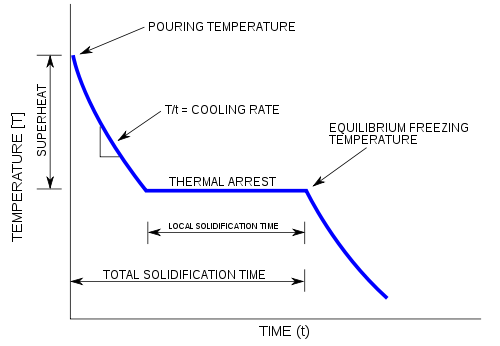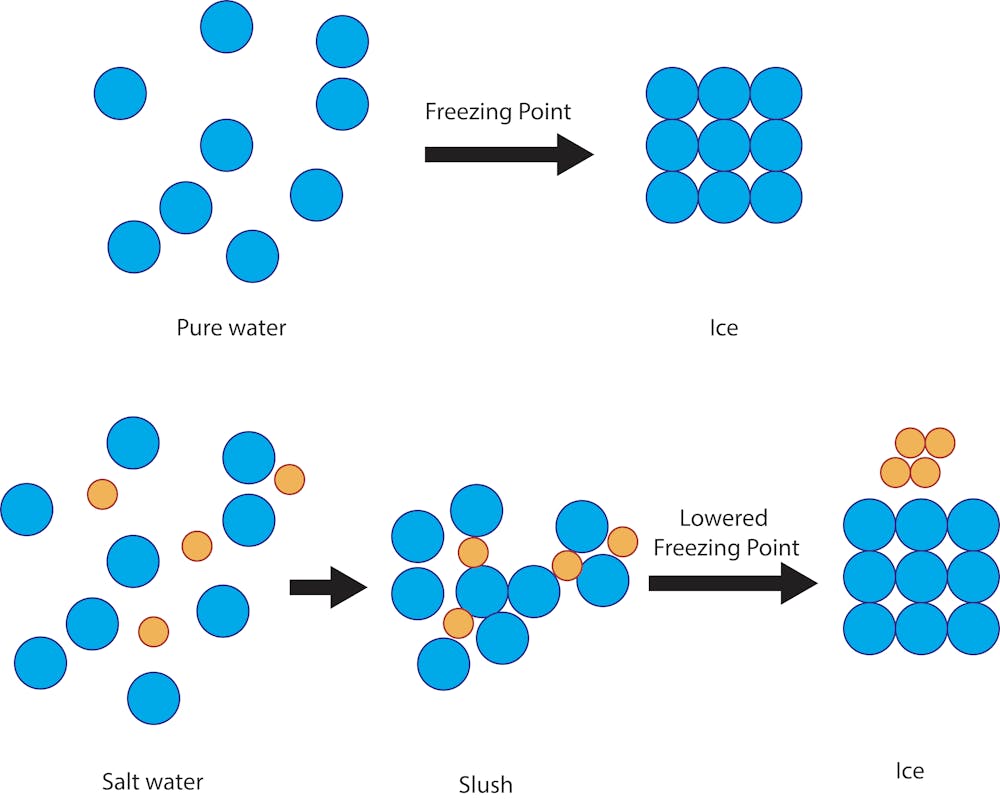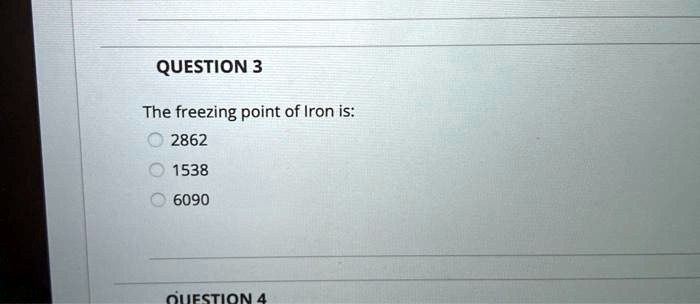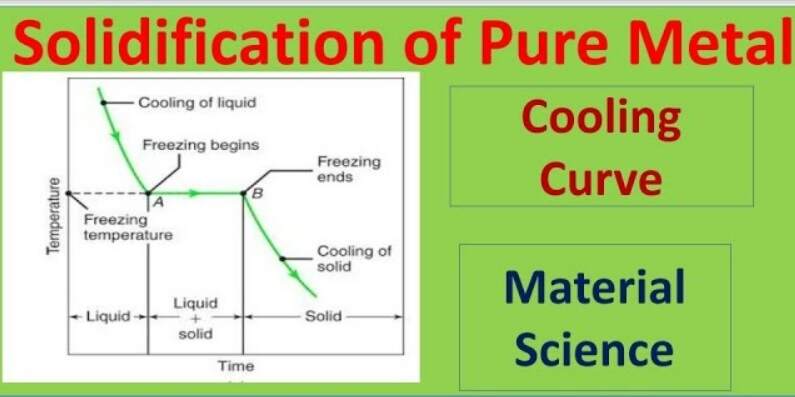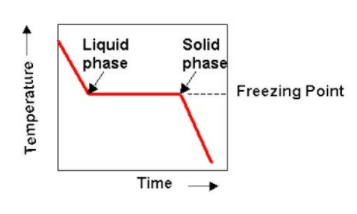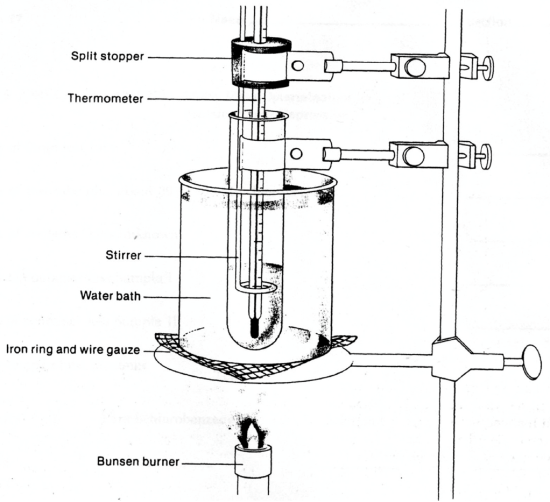
10: Determination of the Molar Mass by Freezing Point Depression (Experiment) - Chemistry LibreTexts
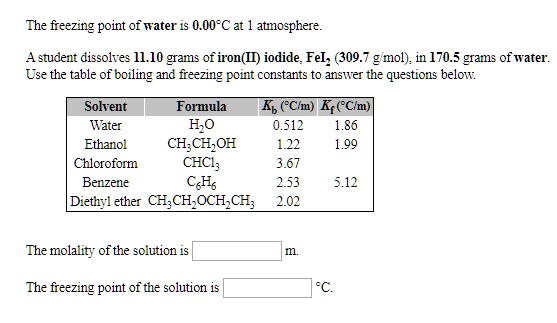
SOLVED: The freezing point of water 0.00-C at atmosphere: Astudent dissolves Il.1O grams of iron(ID) iodide; Fel; (309.7 g mol) in 170.3 grams of water: Use the table of boiling and freezing
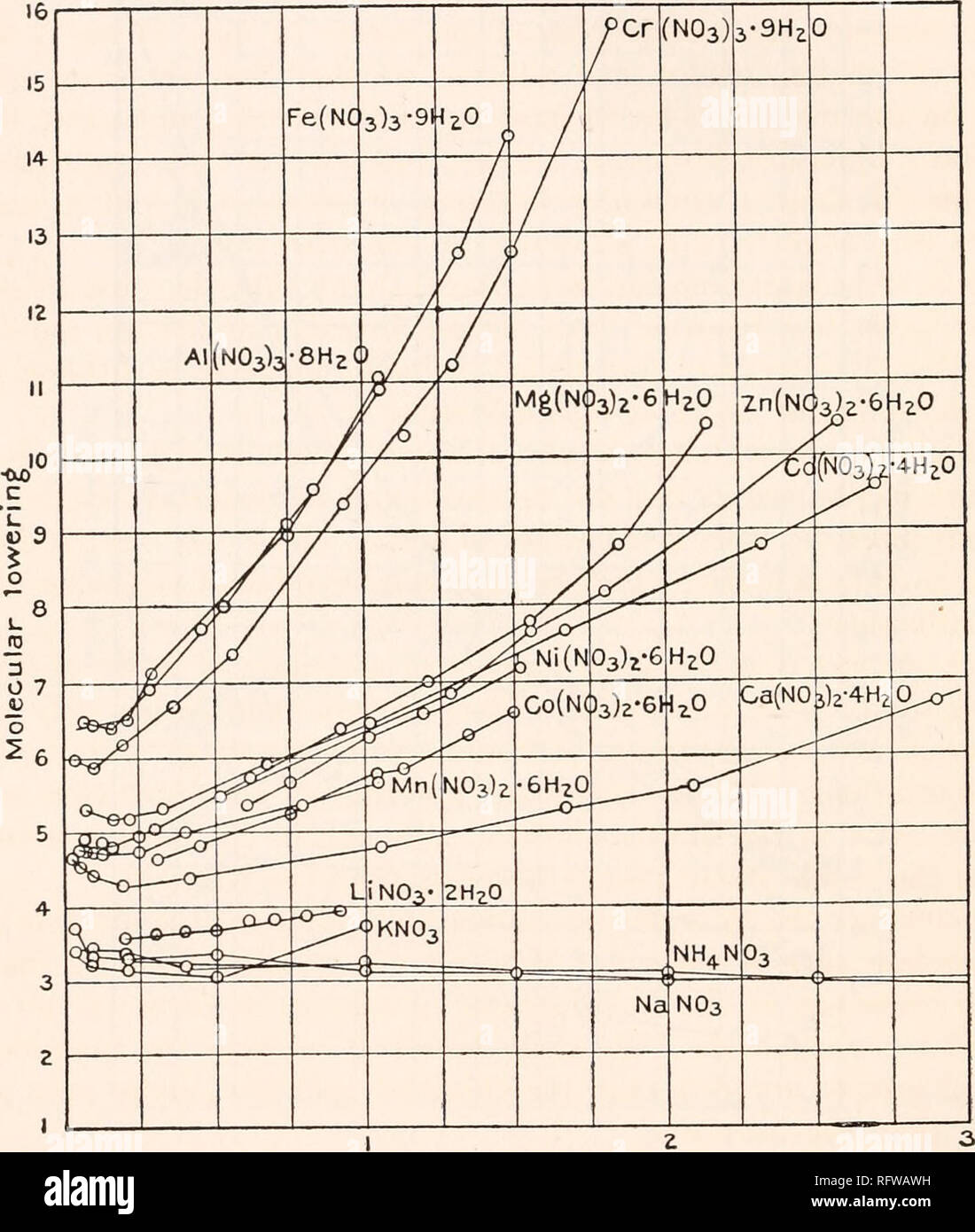
Carnegie Institution of Washington publication. 24 HYDRATES IN AQUEOUS SOLUTION. with this substance, to see if some error had not found its way into this part of the work, and the
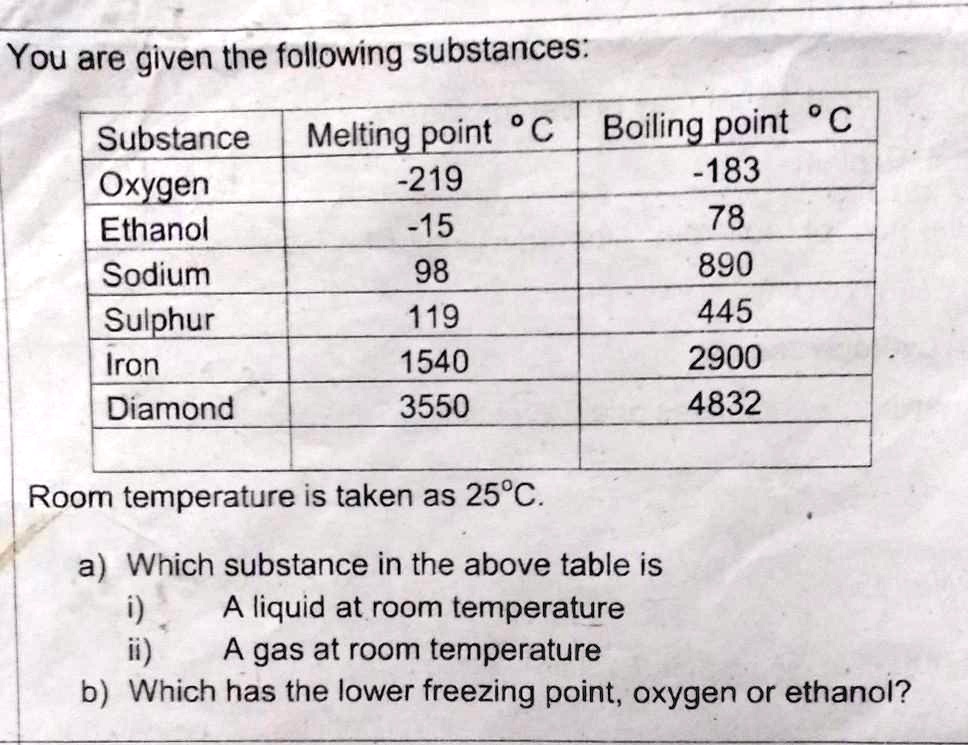
SOLVED: 'give the answer of of this question please You are given the following substances: point C Boiling point C Substance Melting -219 -183 Oxygen Ethanol -15 78 Sodium 98 890 Sulphur

SOLVED: The normal freezing point of certain liquid X is 7.60 %C, but when 101. g of iron(III) nitrate (Fe (NO3) are dissolved in 550- g of X the solution freezes at

Elements Hydrogen Number of: Protons 1 Neutrons 0 Electrons 1 Boiling point -252 Freezing point -259 State at room temperature gas. - ppt download

Boiling point boiling point: the temperature at which a substance changes from a liquid to a gas The BOILING POINT of pure water is 100° Celsius. - ppt download

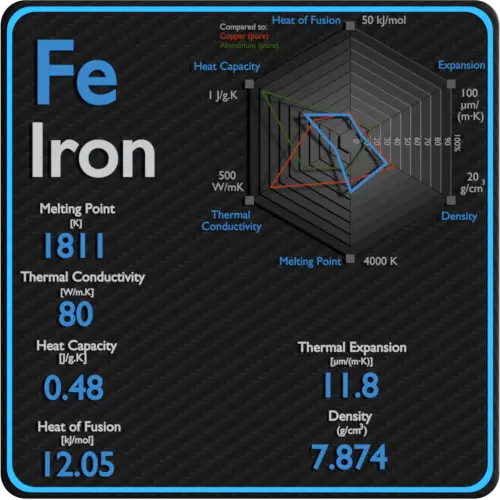


![Iron-Carbon Phase Diagram Explained [with Graphs] Iron-Carbon Phase Diagram Explained [with Graphs]](https://fractory.com/wp-content/uploads/2020/03/Iron-carbon-phase-diagram-explained.jpg.webp)