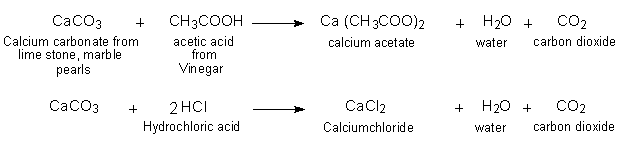
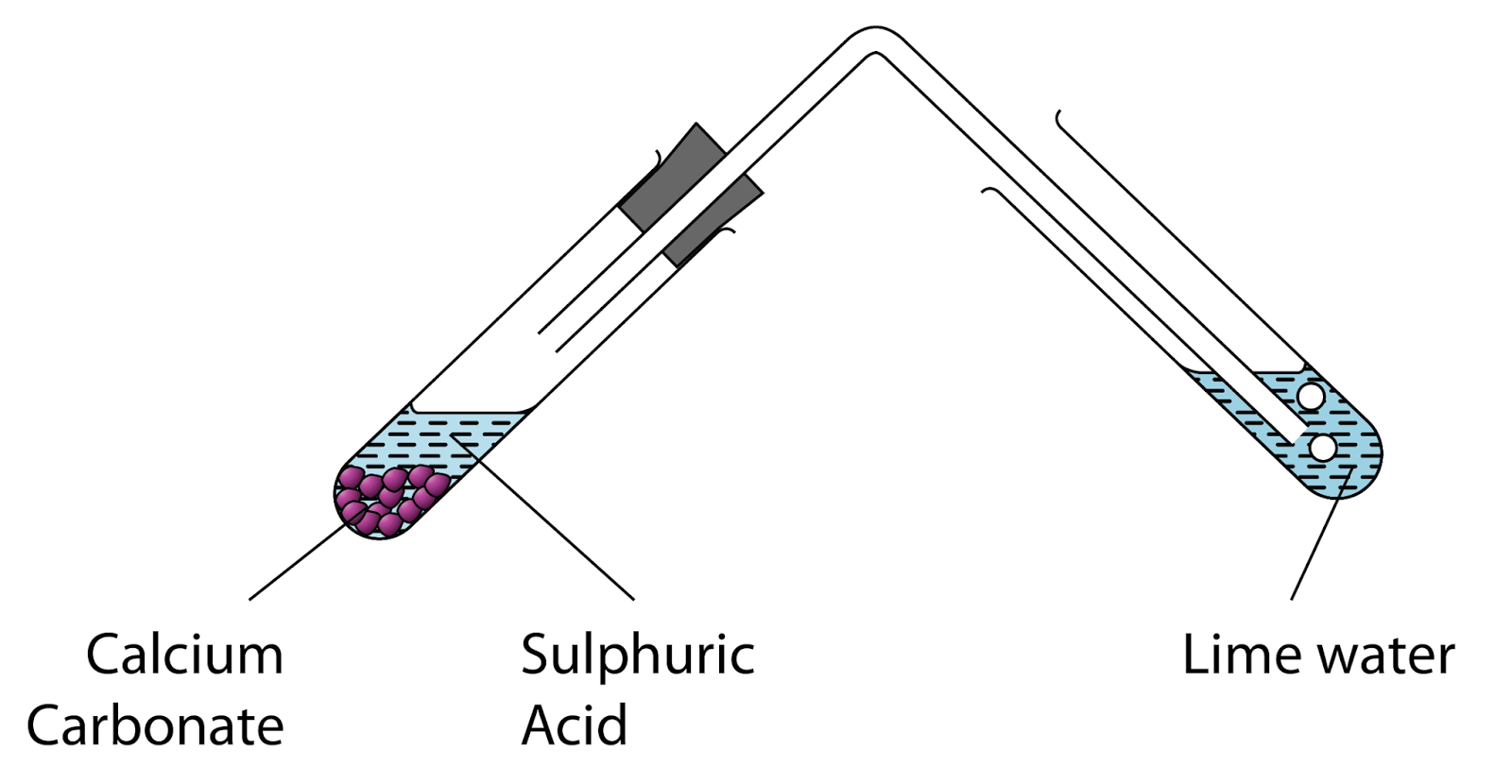
Look figure 4.1 and answer the following question.(a) What change would you observe in the calcium hydroxide solution taken in tube B? (b) Write the reaction involved in test tubes A and

Writing a Net Ionic Equation for the Reaction of Solid Calcium Carbonate with a Hydrochloric Acid Solution



.jpg)





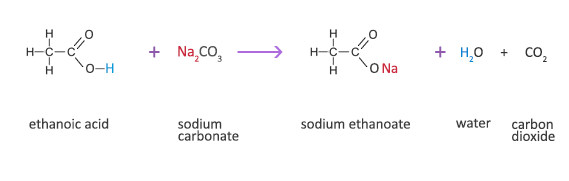
![Carbon] Ethanoic acid - Formation, Properties, Uses [with Reactions] Carbon] Ethanoic acid - Formation, Properties, Uses [with Reactions]](https://d1avenlh0i1xmr.cloudfront.net/fb1b0fbe-d74a-44bb-8d4d-1b41b5aa53a2/reaction-of-ethanoic-acid-with-sodium-carbonate-and-sodium-bicarbonate---teachoo.jpg)
![MCQ] When sodium hydrogen carbonate is added to ethanoic acid a gas MCQ] When sodium hydrogen carbonate is added to ethanoic acid a gas](https://d1avenlh0i1xmr.cloudfront.net/65ccd703-8697-4cd2-9fd1-2b1155ff736e/reaction-of-sodium-hydrogen-carbonate-with-ethanoic-acid---teachoo.png)