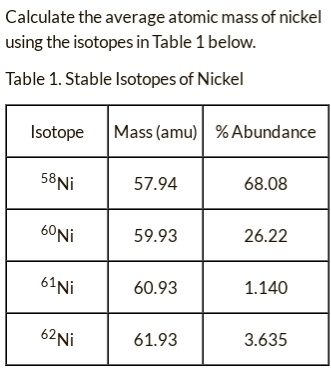
SOLVED: Calculate the average atomic mass of nickel using the isotopes in Table 1 below: Table 1. Stable Isotopes of Nickel Isotope Mass (amu) % Abundance 58Ni 57.94 68.08 60Ni 59.93 26.22 60.93 1.140 62Ni 61.93 3.635 61Ni

Atomic masses L.O.: Define the terms relative isotopic mass and relative atomic mass, based on the 12C scale; Calculate the relative atomic mass of. - ppt download

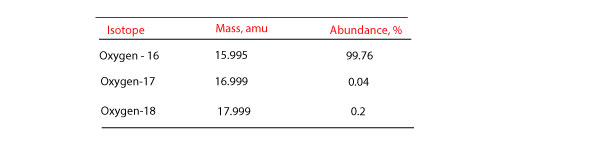
Naturally occurring iron consists of four isotopes with the abundances indicated here. From the masses and relative abundances of these isotopes, calculate the atomic weight of naturally occurring iron. | Homework.Study.com

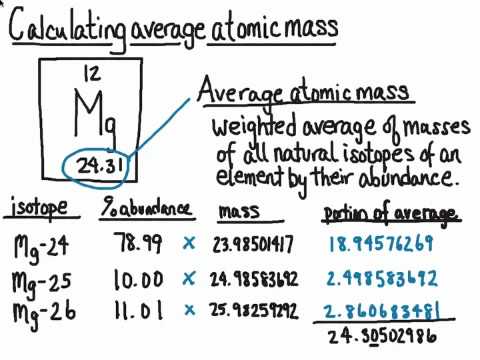
Average Atomic Mass In nature, most elements are a mixture of different isotopes The mass of a sample of an element is a weighted average of all the isotopes. - ppt download
The table below show the relative atomic masses and the percentage abundance of the isotopes L1 and - Tutorke