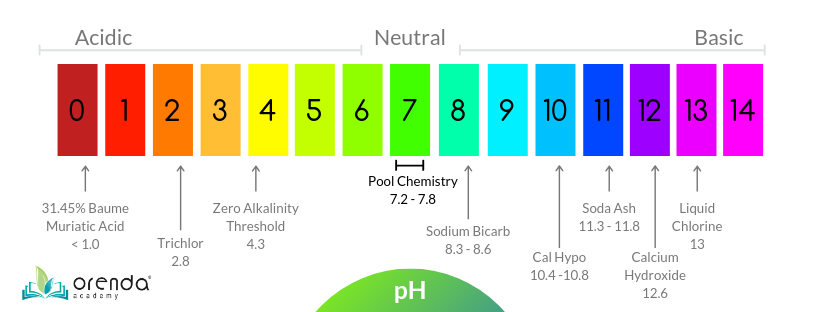
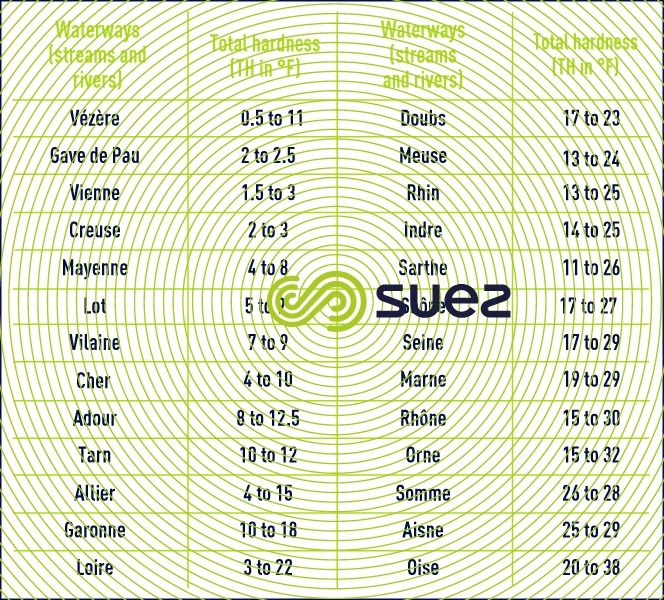
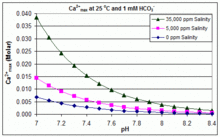
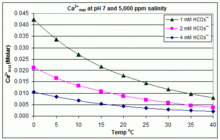
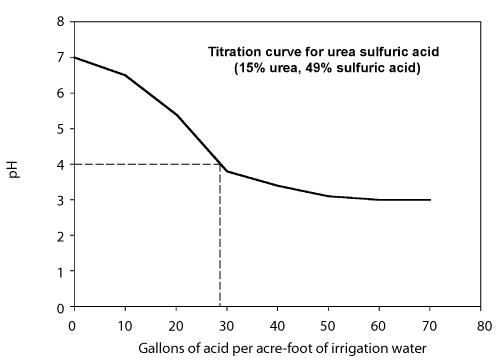
Uncovering the Role of Bicarbonate in Calcium Carbonate Formation at Near‐Neutral pH - Huang - 2021 - Angewandte Chemie International Edition - Wiley Online Library

Figure 2 from EFFECTS OF pH ON CALCIUM CARBONATE PRECIPITATION UNDER MAGNETIC FIELD | Semantic Scholar

Solubility of calcium carbonate (lime scale) in water as a function of pH. | Download Scientific Diagram
What is the pH reading after the endpoint of the neutralization reaction of calcium carbonate and HCL? - Quora

Figure 6 from EFFECTS OF pH ON CALCIUM CARBONATE PRECIPITATION UNDER MAGNETIC FIELD | Semantic Scholar
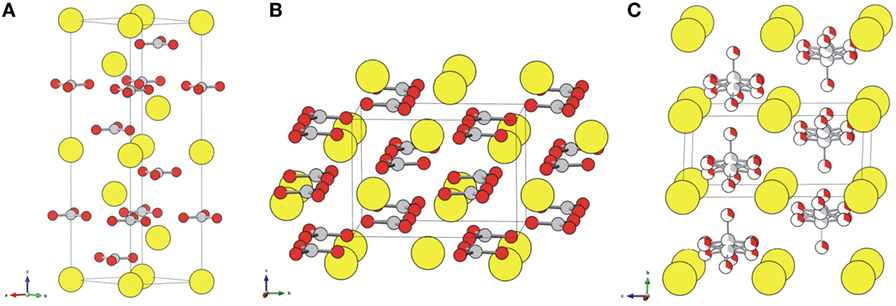
Frontiers | Calcium Carbonate Precipitation for CO2 Storage and Utilization: A Review of the Carbonate Crystallization and Polymorphism

Effect of pH and Phosphate on Calcium Carbonate Polymorphs Precipitated at near-Freezing Temperature | Crystal Growth & Design

Figure 3 from EFFECTS OF pH ON CALCIUM CARBONATE PRECIPITATION UNDER MAGNETIC FIELD | Semantic Scholar

Effect of pH and temperature on calcium carbonate precipitation by CO2 removal from iron‐rich water - Korchef - 2020 - Water and Environment Journal - Wiley Online Library

Effect of pH and Phosphate on Calcium Carbonate Polymorphs Precipitated at near-Freezing Temperature | Crystal Growth & Design

Influence of pH on morphology, size and polymorph of room temperature synthesized calcium carbonate particles - ScienceDirect